| J Korean Neurol Assoc > Volume 44(1); 2026 > Article |
|
Abstract
Cerebrospinal fluid (CSF) dynamics are increasingly recognized as central to brain waste clearance. Beyond the classical view of unidirectional CSF circulation, recent studies have identified the glymphatic system and meningeal lymphatic vessels as major complementary pathways. Glymphatic transport is driven by arterial pulsation, respiration, vasomotion, and strongly enhanced during slow-wave sleep. Advances in neuroimaging, including DTI-ALPS, EPVS quantification, contrast-enhanced magnetic resonance imaging (MRI), and ultrafast functional MRI, have enabled in vivo assessment of these mechanisms. Impaired glymphatic function has been linked to neurodegenerative and cerebrovascular diseases, highlighting its clinical relevance. Potential interventions range from lifestyle measures such as sleep optimization, exercise, and vascular risk control, to pharmacological and experimental strategies aiming to modulate CSF flow. These insights open new opportunities to target the glymphatic-lymphatic axis for the prevention and treatment of neurological disorders.
뇌척수액(cerebrospinal fluid, CSF)은 뇌를 물리적으로 보호하고 대사 노폐물과 신경전달물질을 제거하는 역할을 한다. 고전적인 교과서 모델에 따르면 CSF는 주로 뇌실 내 맥락총(choroid plexus)에서 분비되어 뇌실을 거쳐 지주막하강으로 순환하며 하루 약 400-600 mL가 생성된다[1]. 이후 뇌와 척수를 둘러싼 지주막하강의 CSF는 두개강 정맥동으로 돌출된 지주막 융모(arachnoid villi)를 통해 정맥혈로 흡수된다고 이해되어 왔다[1]. 그러나 이러한 단방향의 CSF 순환 및 흡수 모델은 일부 제한점이 지적되어 왔다. 우선 수두증 모델 등에서 맥락총 이외의 CSF 생성원이 존재할 가능성이 제기되었다[2]. 실제로 아쿠아포린-1 (aquaporin-1, AQP1) 수로 단백질 유전자 결손 생쥐에서도 CSF 생산은 정상 대비 20-30%만 감소하여 맥락총 이외 경로로의 수분 이동이 상당함을 시사하였다[3]. Bulat-Klarica-Orešković 등은 뇌실질 모세혈관이 CSF를 지속적으로 생산하고 흡수한다는 새로운 가설을 제시하여 뇌실질의 사이질액(interstitial fluid, ISF)과 뇌척수액 사이에 상시적인 체액 교환이 일어난다고 주장하였다[4]. 또한 지주막하강에서의 CSF 흡수가 오직 지주막융모만을 통해 이루어지는지는 의문으로 남아 있었다. 실제로 전통적으로 주요 CSF 배출 통로로 여겨졌던 지주막융모의 일방향 밸브설은 전자현미경 관찰에서 완전한 내피장벽이 존재함이 확인되면서 재고되었고 생체 내에서 이들의 역할을 직접 입증한 확실한 근거도 부족하다[5].
지난 수십 년간의 연구는 이러한 고전설을 보완하는 뇌 글림파틱(glymphatic) 계통과 수막 림프관 경로의 존재를 밝혀냈다. 글림파틱 계통은 2012년 뇌조직 내에 림프관이 없다는 점을 고려하여 제창된 개념으로 ‘신경아교세포(glia)’와 ‘림프(lymphatic)’의 합성어이다[6]. 이는 CSF와 ISF 사이의 교환 및 노폐물 제거를 담당하는 새로운 유체 순환 경로로 주변 말초 조직의 림프 배액 기능을 뇌 내에서 부분적으로 대행하는 것으로 이해된다. 한편 2015년에는 지주막공간을 따라 존재하는 뇌수막 림프관이 설치류에서 처음 확인되었고[7] 이후 인간을 포함한 포유류 뇌막에도 림프관이 존재하여 CSF 배출을 돕는다는 사실이 일련의 연구를 통해 재발견되었다[8-10]. 특히 뇌 기저부의 수막 림프관은 배측부(dorsal)보다 구조적으로 발달되어 있고 CSF 흡수에 유리한 위치에 있는 것으로 보고되었다. 2019년 Ahn 등[11]은 두개저 기저부의 수막 림프관이 CSF 배출의 핵심 통로임을 동물 실험으로 증명하였고 2024년 Yoon 등[12]은 설치류 모델에서 비인두 림프관 얼기(nasopharyngeal lymphatic plexus)가 깊은 경부 림프절로 향하는 CSF 유출의 중추적 허브임을 밝혔다. 이러한 발견들은 CSF가 단순히 정맥혈로만 흡수되는 것이 아니라 뇌 주변부의 여러 림프 경로를 통해 다원적으로 배출됨을 시사한다. 본 논문에서는 이와 같이 급격히 진화한 CSF 역동학에 대한 새로운 개념들을 정리하고 글림파틱 계통의 해부생리 기전과 뇌질환에서의 기능적 중요성을 최신 지견에 근거하여 종합적으로 고찰하고자 한다.
고전적으로 CSF의 대부분은 뇌실막의 맥락총에서 능동적으로 분비된다고 여겨졌다. 맥락총은 모세혈관 주위에 특화된 상피세포층으로 구성된 조직으로 혈액뇌척수액장벽(blood-CSF barrier)의 핵심 요소이다[13]. Na+/K+ ATPase 및 이온수송체의 작용으로 혈장으로부터 뇌실 내로 Na+와 염소 이온이 이동하고 이로 인한 삼투 기전으로 수분이 AQP1 수로를 통해 따라 들어와 CSF를 형성한다는 것이다. 하지만 이 모델만으로는 전체 CSF 생성량을 설명하기 어렵다는 문제가 존재한다. 앞서 언급한 것과 같이 AQP1유전자를 결손시켜 맥락총의 수분 이동을 5배 가량 감소시켜도 CSF 생산 감소는 약 20-35%에 그쳤다[3]. 이는 맥락총 이외의 뇌실질 및 모세혈관 기원 CSF 생성이 상당함을 시사한다. 실제로 확산-침투 이중 기전 모델에 따르면 뇌 모세혈관을 통해 혈장 성분이 지속적으로 ISF로 유출되고 이 일부가 뇌실이나 지주막하강으로 진입하여 CSF로 기능한다는 개념이 제안되었다[14,15]. Klarica 등[16]은 이러한 혈관-뇌실질-지주막 하강의 연속적 체액 교환이 CSF 순환을 이루며 CSF 생성과 흡수가 국소적으로 항상 일어난다고 주장하였다.
전통 모형에서는 맥락총에서 생성된 CSF가 측뇌실에서 제3뇌실, 제4뇌실, 지주막하강 방향으로 일방향 대류(convection) 흐름을 타고 이동한 뒤 척수 지주막하강과 두개강 뇌바닥수조(basal cistern)를 거쳐 대뇌 볼록면(cerebral convexicy) 방향으로 순환한다고 설명하였다. 이러한 순환에는 뇌실맥동에 의한 정수압 차가 원동력으로 고려되었다[17,18]. 그러나 현대 연구들은 CSF의 움직임이 보다 복잡하고 양방향성일 수 있음을 시사한다. 심장 박동에 따른 뇌혈류 변화로 뇌실과 지주막하강의 CSF가 일시적으로 뇌 쪽/꼭대기 쪽으로 밀려났다 다시 들이치는 맥동성 흐름이 존재하며 호흡 주기에 따라 흉강 내압 변화로 정맥 귀환과 경막정맥 총압이 변동하면서 두개강과 척수강으로의 양방향성 CSF 흐름도 관찰되었다[19-21]. 따라서 CSF 순환은 단순 중력이나 정수압에 의한 일방향 흐름보다는 심박과 호흡에 동조된 복합적 운동으로 파악되고 있다. 아울러 미세혈관의 수축/이완(혈관 운동성)이나 뇌파와 연관된 느린 파동도 CSF 이동의 원동력으로 제안되는 등 자율신경 및 신경 활동과 CSF 역동학의 연관성이 주목받고 있다[22-24].
전통적인 CSF 흡수 경로는 지주막하강의 CSF가 지주막과립(arachnoid granulation)을 통해 시상정맥동(sagittal sinus)으로 배출되는 것으로 설명되었다. 지주막과립은 지주막이 정맥동 내로 돌출된 구조물로 CSF와 정맥혈을 얇은 내피세포 한 층만을 사이에 두고 접하게 하여 압력 차에 따라 CSF가 정맥으로 여과될 수 있게 한다고 여겨졌다. 그러나 이 경로만으로는 모든 상황의 CSF 배출을 설명하기 어렵고 앞서 언급된 대로 미세구조적으로 일방 밸브 기능을 하는지는 논쟁이 있다[5]. 현대 연구들은 다양한 우회 배출로를 밝혀냈는데 대표적으로 코 주변의 체판(cribriform plate)을 통한 경로가 있다. 이는 후각신경을 따라 코 점막 주변 림프총으로 CSF가 흡수되어 비인두 림프관 얼기에서 심부 경부 림프절로 이어지는 경로이다[12,25]. 또한 두개강 경막의 림프관이 최근 중요 경로로 부상하였다. 2015년 Louveau 등[7]은 설치류의 경막정맥동 주위에 존재하는 림프관이 CSF의 일부를 흡수함을 밝혔고 이어 2017-2020년의 임상 연구들은 조영제 주입 자기공명영상(magnetic resonance imaging, MRI)을 통해 인간 경막에서도 정맥동 주변으로 CSF 조영제가 퍼져나가고 경부 림프절로 배출됨을 시각화하였다[8,10]. 특히 Ringstad와 Eide [10]는 요추천자로 투여한 가돌리늄조영제가 수 시간 내 사람 경막정맥동 주변 경막에 축적되는 것을 보고하여 인간 뇌에서도 수막 림프계가 기능함을 증거로 제시하였다. 결과적으로 현재의 CSF 배출 모델에서는 (1) 뇌신경 주위 경로(특히 후각신경[제1뇌신경] 경로가 주된 역할), (2) 경막 수막 림프관 경로, (3) 정맥동 지주막과립 경로의 세 가지가 주요 통로로 받아들여지고 있다. 이 중 설치류에서는 경부 림프절로 이어지는 림프 경로가 지배적이라는 보고가 있으며[12] 인간 역시 지주막과립의 역할이 어느 정도인지는 불분명하여 림프 경로의 기여가 재평가되고 있다.
글림파틱 계통은 Nedergaard 연구진이 2012-2013년 일련의 동물 실험에서 처음 기술한 개념으로 CSF와 ISF 사이의 뇌 내 특수 체액 통로를 의미한다[6]. 이 경로에서는 CSF가 동맥주위 공간을 따라 뇌실질로 유입되고 ISF와 섞여 노폐물을 수거한 후 정맥주위공간을 따라 다시 뇌 주변 CSF 공간으로 배출된다. 글림파틱 계통이라는 명칭은 별아교세포(astrocyte)의 수발 단백질인 아쿠아포린-4 (aquaporin-4, AQP4)에 의존한다는 점과 말초 림프 계통(lymphatic system)처럼 체액과 노폐물 운반 기능을 한다는 점에 착안한 것이다. 글림파틱 경로의 5단계 과정은 다음과 같다.
(1) 동맥 주위 유입: 지주막하강의 CSF 일부가 뇌동맥 혈관 주위공간(perivascular space, PVS)을 통해 뇌실질로 흘러들어 간다. 이때 동맥벽의 박동성이 액체를 밀어 넣는 펌프 역할을 한다.
(2) 뇌실질 내 확산: 유입된 CSF는 미세한 별아교세포 발돌기 끝(feet) 사이의 틈으로 뇌실질에 퍼져나가며 이 과정에서 AQP4 수로가 중요한 통로를 제공한다. AQP4는 주로 혈관 주위의 별아교세포족(endfeet)에 극성화되어 발현되는데, 이 수로를 통한 물 이동이 없으면 CSF-ISF 교환이 현저히 줄어든다. 실제로 AQP4유전자 결손 생쥐에서는 뇌 내 베타-아밀로이드 등이 효과적으로 제거되지 못하여 글림파틱 흐름이 저하됨이 관찰되었다[6]. 또한 AQP4유전자 결손 생쥐에서 알파-시누클레인 제거가 감소되는 것이 보고되었다[26].
(3) ISF 혼합 및 노폐물 제거: 뇌실질 내로 들어온 CSF는 기존의 ISF와 섞여 확산되며 이때 대류성 흐름(convection)과 확산(diffusion) 작용이 함께 일어난다. 비교적 큰 분자와 노폐물은 PVS나 백질 경로를 따라 비교적 빠르게 이동하는 벌크 흐름에 실려 나가며 작은 분자는 주변 조직으로의 확산에 더 크게 의존한다. 이 과정에서 뇌세포 대사산물(베타-아밀로이드, 타우 단백질, 젖산 등)이 CSF로 씻겨 나오게 된다.
(4) 정맥주위 배출: 노폐물을 실은 CSF/ISF는 뇌 정맥주위공간을 따라 뇌 밖으로 배출된다. 특히 뇌 깊은 구조의 백질을 관통하는 정맥이나 배액정맥을 따라 ISF가 이동하여 최종적으로 경막정맥동 주변의 지주막하공간(뇌바닥수조[basal cistern] 등)으로 모인다. 이를 정맥주위 배출(peri-venous drainage)이라고 하며 뇌의 대사 노폐물이 주로 이 경로로 빠져나오는 것으로 알려져 있다.
(5) 뇌 밖으로의 최종 배출: 정맥 주위로 배출된 CSF-노폐물 혼합액은 이후 뇌막의 림프관으로 흡수되어 깊은 경부 림프절로 이어지거나 일부는 지주막과립을 통해 정맥혈로 직접 들어간다. 또 일부는 두개신경 및 척수신경 주변의 경막초를 타고 나가 주변 림프망으로 유입된다. 즉 글림파틱 계통이 뇌 내 1차적인 배액 경로라면 수막 림프계와 기타 경로는 2차 배출 통로로 기능하여 노폐물을 체외로 최종 이송하게 된다.
글림파틱 계통에서 뇌실질 유체 교환을 가능케 하는 핵심 요소는 별아교세포의 발달이다. 별아교세포의 말단 돌기(process)들은 뇌 미세혈관과 신경세포 사이의 사이질공간을 둘러싸고 있으며 특히 혈관 주위에서는 밀착 돌기를 형성하여 모세혈관 벽을 감싼다. 이 부위 세포막에는 수분통로단백질인 AQP4가 고밀도로 발현되어 있는데 이를 AQP4 극성 분포라고 한다[27]. 이러한 배열 덕분에 PVS로 들어온 CSF의 수분과 용질이 빠르게 뇌실질로 퍼질 수 있다. 초기 연구에서 AQP4를 삭제하면 동물의 뇌 내 베타-아밀로이드 배출이 55% 감소하고 CSF-ISF 혼합이 현저히 저하되어 글림파틱 기능이 거의 정지함을 보였다[6]. 따라서 AQP4는 글림파틱 흐름의 필수적 매개자로 AQP4의 기능 이상이나 배열 변화는 곧 글림파틱장애로 이어질 수 있다. 실제로 노화 쥐 모델에서 별아교세포의 AQP4 극성이 소실되고 주변 신경 염증이 증가하면서 노폐물 배출 효율이 떨어진다는 보고가 있으며[28] 인간 알츠하이머병 환자 뇌에서도 AQP4 극성 분포가 교란된 소견이 관찰되었다고 한다[29].
글림파틱 경로가 활용하는 해부학적 통로는 주로 PVS이다. 동맥이나 정맥이 뇌 조직을 관통할 때 혈관 주위를 둘러싼 미세한 공간이 존재하며 이는 일종의 ISF 통로 역할을 한다. 젊고 건강한 뇌에서는 PVS의 크기가 매우 작아 일반 MRI로는 보이지 않지만 노화나 혈관성 질환에서 이 공간이 확장되어 확장된 혈관주위공간(enlarged PVS, EPVS)으로 나타난다[30,31]. PVS는 위치에 따라 분류되며 기저핵 주위의 렌즈핵줄무늬체동맥(lenticulostriate artery)을 따른 기저핵형(type I), 뇌백질 내 관통 소동맥 주변의 난형 중심형(type II), 중뇌 주위 등에서 드물게 관찰되는 중뇌형(type III) 등이 있다[32]. EPVS는 질환 연관성에 차이가 있는데 기저핵 부위 EPVS는 고혈압, 노화, 소혈관 질환(열공뇌경색)과 연관이 크고 반난형 백질중심부(centrum semiovale)의 EPVS는 재발성 미세출혈이나 뇌아밀로이드혈관병과 연관되며 알츠하이머병 환자에서도 빈도가 높다[30,31,33-35]. 이러한 EPVS를 MRI에서 수, 크기, 체적 등을 정량화하거나 시각적으로 등급화하여 뇌 노폐물 배출 및 신경퇴행질환 진행을 가늠하는 간접 지표로 활용하려는 시도가 있다.
글림파틱 흐름을 움직이는 동력으로 몇 가지 생리 요인이 제안된다. 첫째는 뇌맥락총의 지속적 CSF 생산이다. 일정한 속도로 뇌실 내 CSF가 만들어져 밀려나오므로 이것이 글림파틱 유입의 기본 압력 구배를 제공한다. 둘째, 동맥 박동이 중요하다. 동맥벽이 수축기마다 팽창하며 PVS를 압박함으로써 CSF가 조직으로 밀려들고 확장기에는 이완되며 액체를 끌어들이는 펌프 역할을 한다. Iliff 등[6]의 연구에서 두광자 현미경 연구는 동맥 맥동이 없으면 뇌사이질로 조영액 유입이 감소함을 직접 확인하였다. 셋째, 호흡 및 심박 변동이다. 호흡으로 인한 흉강압 변화가 경정맥 유출과 두개강 내 압력을 주기적으로 변동시키며 이 압력파가 CSF를 상하로 흔드는 큰 동인이 된다. MRI로 측정한 CSF 유속 신호에서 호흡 주파수 성분이 심박 성분보다도 크게 나타나 호흡 리듬이 CSF 순환에 미치는 영향이 강조되기도 하였다. 넷째, 자율신경성 혈관 운동(vasomotion)이다. 뇌실질세동맥의 지배 근육이 교감/부교감신경 신호에 따라 지름 변화를 일으키는 저주파 혈관 운동은 분당 수 회 정도의 느린 맥동을 뇌에 전달한다. 이러한 저주파 혈관 톤 변화 역시 혈관주위액의 이동을 촉진하는 요인으로 거론된다. 마지막으로 수면-각성 상태가 글림파틱 흐름에 큰 영향을 미친다. Xie 등36의 연구에 따르면 동물에서 각성 시보다 수면 또는 마취 상태에서 사이질공간이 60% 이상 확대되고 그 결과 CSF가 뇌조직으로 더 깊이 침투하여 노폐물 제거가 2배 이상 빠르게 일어났다. 이는 수면 중 ISF 순환이 활성화됨을 시사하며 수면 시 교감신경 활성이 감소하여 뇌세포외공간을 수축시키던 노르에피네프린 신호가 줄어드는 것이 기전으로 제시되었다. 종합하면 동맥 박동, 호흡 리듬, 뇌압 변화, 수면 상태 등이 글림파틱 계통의 주요 조절 인자로 작용한다.
글림파틱 경로를 통한 뇌 노폐물 이동에 대류성 흐름(convection)이 얼마나 기여하는지 혹은 단순 확산(diffusion)이 주된 기전인지는 논쟁의 대상이다. Nedergaard 연구진은 비교적 큰 분자들이 비교적 먼 거리까지 빠르게 이동하는 현상을 근거로 벌크 흐름에 의한 실질적 대류가 존재한다고 주장하였다[6,37]. 반면 Smith 등[38]은 뇌조직에서 그렇게 대규모의 체액 흐름이 지속적으로 일어나기 어렵고 실은 실험 조작으로 인한 인공 산물일 수 있으며 확산이 주된 기전이라고 반박하였다. 현재는 대류와 확산이 모두 기여하되 분자 크기와 경로에 따라 비중이 다를 수 있다는 절충적 견해가 유력하다(Table 1) [39,40]. 또한 글림파틱 흐름의 방향성도 논의되고 있다. 원래 개념은 동맥, 뇌실질, 정맥 방향이지만 일부 연구는 동맥벽 내측의 모세혈관 기저막을 통한 동맥벽 내 배출(intramural periarterial drainage) 경로를 제시하며 글림파틱 모델과 대립하기도 한다. 예컨대 Albargothy 등[15]은 베타-아밀로이드가 동맥 기저막을 타고 외부로 이동한다는 관찰을 들어 글림파틱보다는 혈관벽 내 배출이 주 경로라고 주장하였다. 이 역시 현재 활발한 연구와 토론이 진행 중인 분야이다.
수면은 글림파틱 계통 활성에 가장 큰 영향을 미치는 인자로 꼽힌다. 앞서 소개한 Xie 등[36]의 연구에서 제시된 바와 같이 깊은 수면 또는 마취 상태에서는 뇌세포사이공간이 확장되고 노폐물 제거가 가속화된다. 이는 각성 시 교감신경 활성도가 높은 상태에서는 사이질공간이 좁아지고 뇌혈관이 수축하여 글림파틱 유입이 억제되지만 수면, 특히 느린 비렘(non-REM)수면 동안에는 교감신경 활동 저하와 느린 뇌파 발생으로 글림파틱 흐름이 원활해지기 때문이다. 규칙적인 수면 주기는 또한 별아교세포의 AQP4 분포를 유지하고 뇌 대사 항상성을 돕는 것으로 생각된다. 반대로 만성적인 수면 부족이나 수면장애(예: 수면무호흡증)는 글림파틱 노폐물 제거를 저해하여 알츠하이머병 등의 위험을 높일 수 있다는 영상 연구들이 발표되었다[41]. 실제로 야간 수면무호흡증 환자에서 DTI-ALPS 지표가 낮고 뇌 노폐물(베타-아밀로이드 등) 축적이 높은 경향이 보고되었으며 양압 환기 치료로 수면의 질이 개선되면 이러한 지표도 호전될 가능성이 제기되었다[42,43]. 일주기(circadian) 리듬 또한 글림파틱 기능에 관여하는데 낮에는 글림파틱 흐름이 상대적으로 떨어지고 밤에 상승하는 양상이 동물 실험에서 관찰되었다[44]. 이는 뇌 내 시계유전자가 뇌세포 대사와 이온 균형을 조절함으로써 간접적으로 유체역학을 변화시키는 것으로 추정된다.
자율신경계의 교감-부교감 주기는 뇌혈관 직경과 CSF 생성 속도 등에 영향을 미친다. 예를 들어 낮 시간 스트레스나 교감신경 항진 상황에서는 뇌동맥이 수축하고 맥락총 혈류가 감소하여 CSF 생산이 줄어들며 뇌실질의 글림파틱 유입도 억제될 수 있다. 반대로 부교감신경이 우세한 안정 상태에서는 뇌혈류가 늘고 CSF 생성이 늘어 글림파틱 순환을 촉진하는 경향이 있다. 이러한 효과는 수면-각성 주기와도 상당 부분 겹치지만 각성 상태에서도 명상이나 이완 등으로 부교감신경 활성도가 높아지면 일시적으로 글림파틱 순환이 증진될 수 있다는 가설이 있다[45]. 동물 연구에서는 교감신경 차단제를 투여하거나 경동맥 신경총을 절단하여 교감 긴장을 낮추면 ISF 흐름이 증가함이 관찰된 바 있으며 반대로 외상 후 카테콜아민 급증 등의 급성 교감신경폭풍(sympathetic storms)은 글림파틱 유입을 거의 완전히 차단하기도 하였다[46]. 부교감신경계 중 미주신경의 역할도 거론되는데 미주신경 자극이 뇌혈류 개선을 통해 글림파틱 기능을 높일 가능성이 논의되고 있다[47].
호흡은 심장 박동보다도 더 큰 주기적 압력 변화를 야기하여 CSF 순환을 주도한다. 심호흡을 할 때마다 두개강과 척수강 사이로 CSF가 출렁이는 현상은 20세기 중반부터 알려졌는데 최근 Kiviniemi 등[48]은 사람에게서 기능 MRI (functional MRI, fMRI)로 심박과 호흡의 CSF 박동 기여도를 분석하여 호흡 박동이 심장 박동보다 더 강하게 기여함을 보고하였다. 이는 느린 호흡운동이나 일시적 과호흡/무호흡 등이 CSF 흐름에 상당한 영향을 줄 수 있음을 의미한다. 실제로 일부 연구는 수면 중 일어나는 깊은 호흡이나 한숨 등이 대뇌 blood oxygenation level dependent (BOLD) 신호와 동반되어 CSF 상승/하강을 유발한다고 하였다[24]. 이러한 발견을 바탕으로 호흡을 의식적으로 조절하여 글림파틱 순환을 증진시키는 방법에 대한 관심도 생겨났다. 예를 들면 요가 호흡이나 호흡 완화 요법 등이 글림파틱 기능에 도움을 줄 수 있는지에 대한 임상시험이 시작되고 있다(NCT06989242) [49].
혈압과 맥압의 크기, 심박수 등은 PVS의 압력 파동에 영향을 미친다. 앞서 기술한 대로 동맥의 맥동 자체가 글림파틱 유입의 추진력 중 하나다[50]. 그러므로 저혈압 또는 동맥경화로 맥압 전파가 감소하면 글림파틱 순환이 둔화될 수 있고[51] 반대로 고혈압이나 맥압 항진 상태에서는 처음에는 글림파틱 유입을 늘릴 수 있으나 만성적으로는 혈관 탄성 저하와 혈뇌장벽(blood-brain barrier, BBB) 손상을 불러와 오히려 방해가 될 수 있다[52]. 결국 적절한 심뇌혈관 건강 유지가 뇌 글림파틱 건강에도 중요하며 심질환자에서 인지기능 저하가 흔한 이유 중 하나로 이러한 기전이 연구되고 있다[53].
글림파틱 및 뇌수막 림프 계통의 기능을 인체에서 비침습적으로 평가하기 위하여 다양한 영상 기법과 생물표지자가 연구되고 있다. 최근 5-10년간 개발된 대표적 방법들을 요약하면 다음과 같다.
CSF 공간에 가돌리늄조영제를 주입하여 시간 경과에 따른 분포를 추적하는 방법이다. 수막 내(intrathecal) 경로(요추천자를 통한 천막하강 주입)로 조영제를 투여한 후 연속 MRI 촬영을 통해 뇌실질 및 수막으로의 확산 양상을 관찰하면 글림파틱 순환의 역동학을 파악할 수 있다[37]. 실제로 Ringstad 등[54]은 이 방법으로 정상인과 수두증 환자의 CSF 순환 차이를 정량화하여 보고하였다. 최근에는 정맥 주사한 조영제가 BBB를 통과하지 못하므로 경막 림프에만 선택적으로 모이는 현상을 이용하여 정맥 내 경로 조영증강으로 수막 림프관을 간접 평가하려는 시도도 있다[8].
확산텐서영상(diffusion tensor image, DTI)을 이용한 뇌혈관 주위 확산률 분석(analysis along the perivascular space, ALPS) 지표는 2017년 Taoka와 Naganawa가 제안한 기법이다[55]. 뇌 내 특정 백질로(fornix 주변 등)에서 확산 이방성의 차이를 측정함으로써 PVS를 따라 일어나는 ISF 확산의 상대적 용이도를 지표화한 것이다. ALPS 지표가 높을수록 글림파틱 순환을 통한 노폐물 제거 능력이 양호한 것으로 간주되며 정상 노인 대비 알츠하이머병 치매 환자 등에서 ALPS 지표가 유의하게 낮다는 연구들이 다수 보고되었다[8,56,57]. 국내 한 연구에서는 파킨슨병 환자에서 ALPS 지표 감소 및 운동/인지기능 저하의 상관관계를 발표하였고[58] 낮은 ALPS 지표가 파킨슨병의 장기 예후와 연관된다는 연구 결과가 보고되었다[59]. 당뇨병, 뇌소혈관질환(cerebral small vessel disease, CSVD) 등 다양한 질환에서 이 지표의 유용성이 검증되고 있다[60,61].
그러나 DTI-ALPS 지표는 백질 내 국소 영역의 수분 확산을 측정하는 간접 지표에 불과하여 피질을 포함한 공간적, 시간적으로 다양한 CSF-ISF 교환 및 복잡한 노폐물 제거 정도를 완전히 반영하기 어렵다는 비판도 있다[62]. 피질-백질 PVS의 연속성 가정과 배액해부학(표재, 심부수지정맥 등)에 논란이 있어 해석은 신중해야 한다.
앞서 설명한 대로 MRI에서 PVS의 확장 정도는 만성 뇌노폐물 배출 부전을 반영할 가능성이 있다. 임상 연구에서는 MRI의 T2강조영상이나 액체감쇠역전회복영상(fluid-attenuated inversion recovery imaging, FLAIR)에서 EPVS의 개수를 시각적으로 점수화하거나 자동화 소프트웨어를 통해 전체 부피를 산출하여 글림파틱 건강도의 지표로 활용한다[63,64]. 예를 들어 한 연구에서는 경도인지장애 및 알츠하이머병 치매 환자군이 정상 대조군보다 반난형백질중심부(centrum semiovale) EPVS 부피가 유의하게 크다고 보고하였고[33] 다른 연구에서는 EPVS가 심할수록 아밀로이드 양성과 연관된 혈청 Aβ40/A42 분율이 낮다고 보고하였다[65]. 국내 연구들에서도 경도인지장애 및 알츠하이머병 치매 환자에서 EPVS의 부피와 개수가 증가하고 인지 저하 속도와 연관됨이 보고되었으며[66] 파킨슨병 환자에서도 기저핵 EPVS가 인지 저하, 신경정신 증상, 장기적인 운동 예후와 밀접히 관련됨이 확인되었다[67-69].
최근에는 미세순환과 분자 상호작용을 반영하는 고급 자기공명(magnetic resonance, MR) 기법이 글림파틱 연구에 폭 넓게 도입되고 있다. 예를 들어 화적소내비결집운동(intravoxel incoherent motion) MRI는 미세혈류와 확산 성분을 분리하여 뇌실질 및 PVS의 미세순환 정도를 정량화함으로써 글림파틱 유동의 미세한 대류 성분을 비침습적으로 평가하는 데 활용되고 있다[70]. 또한 화학교환포화전달(chemical exchange saturation transfer) MRI는 수소 결합 기반의 분자 교환 반응과 대사물 농도 변화를 감지할 수 있어 글림파틱 계통을 통한 대사 노폐물 축적 및 제거 과정의 분자적 변화를 시각화하는 새로운 접근법으로 주목받고 있다[71].
사람 뇌수막 림프계를 직접 영상화하는 방법으로는 앞서 언급한 3D-FLAIR 단층 촬영이 있다. 이 기법은 높은 echo time과 적절한 inversion time을 사용하여 단백질 농도가 높은 림프액 신호를 선택적으로 강조하는 방식으로 Albayram 등[9]은 이를 디지털감산림프조영술(digital subtraction lymphangiography)과 병합하여 경막 림프관 및 경부 림프 배출 경로를 비침습적으로 시각화하였다. 또한 척수강 내 가돌리늄조영제를 주입하여 척수 주변 림프 배출로를 추적하거나[10,73] 디지털감산림프조영술 등 핵의학 기법으로 뇌 림프 배출을 확인하려는 연구도 진행되고 있다.
이처럼 다양한 영상 생물표지자들이 개발되어 왔으며 아직 표준화된 임상 지표로 쓰이기에는 검증이 더 필요하지만 글림파틱/림프 기능의 간접 지표로써 잠재력이 기대된다.
뇌 글림파틱 계통의 기능 저하는 여러 신경퇴행질환 및 뇌혈관질환에서 병태생리에 중요한 역할을 하는 것으로 추정된다.
정상 노화만 하더라도 수면의 질이 감소하고 별아교세포의 변화로 글림파틱 유동이 떨어지는데, 이는 나이가 들수록 베타-아밀로이드 등의 뇌 축적이 늘어나는 현상과 맥락을 같이 한다. Nedergaard와 Goldman은 글림파틱 실패는 다양한 치매로 가는 최종 공통 경로가 될 수 있다고 강조하였는데, 이는 수면 장애와 노화, 노폐물 배출 저하, 단백질 응집이 서로 악순환 고리를 이루어 알츠하이머병 등의 발병과 진행을 촉진한다는 개념이다[74]. 실제로 알츠하이머병 동물 모델에서는 글림파틱 기능이 떨어지면 베타-아밀로이드 및 타우 병리가 촉진된다는 보고도 있다[75,76]. 또한 인간 환자 대상 연구에서도 경도인지장애/치매 환자에게서 DTI-ALPS 지표 감소, EPVS 증가, CSF 조영 MRI 지표 저하 등의 글림파틱 기능장애 소견이 꾸준히 확인되고 있다[66,77]. 파킨슨병 등의 다른 신경퇴행질환에서도 알파-시누클레인 등 병리 단백질의 축적에 글림파틱장애가 기여할 수 있다는 증거가 있으며[26] 일부 연구는 파킨슨병 환자에서 글림파틱 지표와 운동/인지 증상의 상관성을 제시한다[68,69].
CSVD와 글림파틱 기능 또한 밀접히 연관된 것으로 보인다. 고령에서 흔한 뇌백질 변성이나 열공경색, 미세출혈 등 소혈관 질환 소견들은 전통적으로 만성 허혈과 BBB장애로 설명되었다. 그러나 최근 연구들은 소혈관질환 환자 뇌에서 EPVS의 광범위한 증가와 글림파틱 대사산물 제거 저하를 관찰하고 글림파틱 부전이 백질 손상의 원인 중 하나일 수 있다고 제안하였다[78]. 소혈관질환 환자에서 인지기능 저하 정도가 EPVS 부피와 상관관계를 보였다는 연구도 있어 미세혈관 변화가 글림파틱 흐름을 방해하여 독성 대사산물이 축적되고 궁극적으로 뇌소량 인지장애로 이어질 가능성이 있다[79]. 또한 뇌졸중 후 발생하는 치매에서도 뇌실질의 대사 노폐물 제거 경로가 손상되어 염증 및 신경퇴행을 촉진한다는 가설도 대두되어 있다[80].
외상 뇌손상(traumatic brain injury, TBI) 분야에서도 글림파틱 계통이 중요한 역할을 한다. 급성 외상 후 뇌부종 발생과 회복에 글림파틱 흐름이 관여하며 외상으로 인한 교감신경 폭풍이 글림파틱 기능을 급격히 억제하여 부종을 악화시킨다는 보고가 있다. 2023년 Hussain 등[46]은 쥐의 뇌손상 모델에서 외상 직후 교감신경 흥분을 억제하는 알파/베타차단제 칵테일(프라조신[prazosin]+아티파메졸[atipamezole]과 프로프라놀롤[propranolol])을 투여하면 글림파틱 배액이 회복되고 뇌부종이 유의하게 감소함을 보여주었다. 또한 해당 처치군에서 인산화 타우 단백 축적과 신경 염증이 완화되어 만성 외상 후 뇌병증으로의 진행이 억제되었다고 보고하였다. 이는 TBI에서 glymphatic-stagnated edema, 즉 글림파틱 정체로 인한 부종이 중요한 병인임을 시사하며 글림파틱 경로를 개선하는 치료의 가능성을 보여준다. 임상적으로도 외상 후 높은 두개내압이나 혈종으로 글림파틱 흐름이 막히면 환자 예후가 나쁘다는 관찰이 있고 반대로 외상 후 수면이나 진정으로 글림파틱 흐름을 촉진하면 신경 손상 회복에 도움이 될 수 있다는 가설이 제기되고 있다.
맥락총은 CSF 생성의 주요 기관일 뿐 아니라 혈액뇌척수액 장벽(blood-CSF barrier)을 이루는 면역 조절의 관문으로써 중추신경계의 면역 관문(immune checkpoint) 역할을 수행한다. 이러한 특성 때문에 맥락총은 오래전부터 신경 염증(neuroinflammation)과 밀접한 연관을 가지는 구조로 주목되어 왔다[81]. 나이가 들거나 퇴행성 뇌질환이 발생하면 맥락총에 다양한 형태적 변화가 나타난다. 예를 들어 기저막의 비후, 모세혈관 밀도 감소 및 상피세포 위축, 섬유화 등이 보고되었으며 이는 맥락총의 CSF 생성 능력 및 물질 운반 능력을 떨어뜨린다[82]. 최근에는 맥락총의 부피 증가(choroid plexus enlargement) 자체가 다양한 신경퇴행질환과 연관된다는 사실이 밝혀지고 있으며 특히 글림파틱(glymphatic) 기능장애가이 관계를 매개한다는 보고가 잇따르고 있다.
Li 등[83]은 MRI 기반 연구에서 맥락총 부피가 증가한 환자일수록 백질 고신호병변(white matter hyperintensity)의 진행이 빠르며 이러한 현상이 글림파틱 기능 저하와 밀접하게 관련된다고 보고하였다. Xu 등[84]은 알츠하이머병 환자에서 맥락총 내 유리수(free-water) 증가가 글림파틱 기능 저하와 상관됨을 제시하였으며 Liu 등[85]은 파킨슨병 환자에서 맥락총 비대가 국소적 글림파틱장애를 매개로 운동 증상 중증도를 높인다고 밝혔다. 이들 연구는 맥락총이 단순한 CSF 생성샘을 넘어 면역 및 노폐물 제거 계통의 통합 조절자로 기능함을 시사한다.
본 연구진이 진행한 국내 연구들에서도 이러한 결과가 일관되게 관찰되었다. 알츠하이머병 연속체 환자에서 맥락총 비대가 아밀로이드 축적 및 인지기능 저하와 밀접히 관련됨이 보고되었으며[86] 파킨슨병 환자군을 대상으로 한 연구에서 맥락총 비대가 인지기능 저하뿐 아니라[87] 도파민 신경 변성과 운동 증상의 정도와도 유의한 상관을 보인다고 보고하였다[87]. 이러한 일련의 관찰은 맥락총의 구조적 변화가 신경퇴행질환의 병리와 글림파틱 기능장애를 매개하는 핵심 연결고리로 작용할 수 있음을 시사한다.
아울러 최근 보고된 연구에서는 노화 쥐 모델에서 맥락총의 대사 활성이 유지되며 CSF 분비 기능이 안정적으로 보존된다는 점을 보여주었다[88]. 이는 맥락총의 기능 저하가 단순한 노화의 결과가 아니라 신경퇴행질환에서의 병리적 자극에 의하여 유도되는 변화일 가능성을 시사한다. 이러한 관찰은 향후 연구에서 질환-특이적 맥락총 변화를 조기 지표나 치료 표적으로 활용할 수 있는 가능성을 제시한다[88]. 위와 같이 맥락총이 뇌 면역 및 노폐물 제거에 있어서도 중요한 역할을 한다는 것이 밝혀지고 있다.
이 외에도 뇌염 및 다발경화증, 수두증, 뇌종양 등에서 글림파틱/림프 경로의 변화가 보고되고 있다. 예를 들어 수두증 환자에서 뇌실 확장이 글림파틱 경로를 압박하여 백질의 노폐물 축적을 초래할 수 있고54 종양 환자에서 종양세포 배출이나 면역세포 침윤 경로로 글림파틱이 관여할 수 있다는 연구들이 있다[89]. 전반적으로 글림파틱 계통은 다양한 신경질환의 병태생리에 교차로처럼 위치하며 이는 향후 진단 및 치료 표적으로써 큰 잠재력을 갖는다는 점에서 관심이 집중되고 있다.
글림파틱 계통의 기능을 향상시켜 뇌 노폐물 제거를 촉진하려는 다양한 치료 전략이 모색되고 있다. 이는 크게 생활 요법이나 물리적 방법 등의 비약물적 중재와 약물을 이용한 약리 중재로 나눌 수 있다.
무엇보다 충분한 수면이 최우선 권고된다. 수면의 질을 개선하고 수면무호흡증 등을 적극적으로 치료하여 깊은 느린파 수면을 늘리는 것이 글림파틱 활성에 가장 기본적인 도움이 된다[36]. 수면 자세도 영향을 미칠 수 있다. 동물 연구에서 똑바로 누운 자세보다 옆으로 누운 자세(측와위)에서 글림파틱 배출이 더 잘된다는 보고가 있어 가능하다면 측면 수면을 권장하기도 한다[90]. 규칙적인 유산소 운동은 뇌 혈류와 전반적 림프 순환을 촉진하여 글림파틱 기능을 향상시킨다는 근거들이 있으며 치매 예방을 위한 생활 습관으로도 운동이 강조된다[91]. 음주 조절 또한 중요하다. 소량의 음주는 일시적으로 혈관을 확장시켜 글림파틱 유동을 늘릴 수 있으나 만성적 과음은 수면 구조를 파괴하고 탈수 및 염증을 초래하여 오히려 글림파틱 기능을 저해한다[92]. 따라서 과도한 알코올 섭취를 피하고 절주하는 것이 좋다. 식이 면에서는 오메가-3 등의 불포화지방산 섭취가 뇌혈관 건강과 염증 완화에 기여하여 간접적으로 글림파틱에 이로울 수 있다는 보고가 있다[93]. 마지막으로 적극적인 만성 질환 관리가 필요하다. 고혈압, 비만, 당뇨, 고지혈증 등은 모두 뇌혈관기능과 염증에 영향을 주어 글림파틱 경로를 손상시킬 수 있으므로[94] 이를 잘 조절하는 것이 뇌 노폐물 제거를 돕는 길이다.
최근 몇 년간 글림파틱 활성화 약물에 대한 연구가 활발하다. 앞서 언급한 대로 교감신경차단제들은 글림파틱 흐름을 개선시키는 효과가 관찰되었다. TBI 동물 모델에서 프라조신+아티파메졸(알파 차단)과 프로프라놀롤(베타 차단)을 병용 투여한 결과 손상 후 글림파틱 기능 부전이 교정되고 신경 손상이 감소하여 교감신경 억제 칵테일이 뇌 외상 환자 치료에 시험되고 있다[46]. 심박수를 증가시켜 뇌 펄스 타일을 높이는 도부타민 같은 약물도 동물 연구에서 CSF 순환을 촉진한 바 있다[50]. 뇌척수액의 삼투압 기전을 이용하여 만니톨, 고장성 식염수 등의 고삼투 용액을 정주하면 일시적으로 CSF 흐름이 빨라지고 뇌사이질액이 확장되어 글림파틱 유입이 늘어난다는 보고가 있다[6,95]. AQP4 수로의 활성을 증가시키는 AQP4 촉진 물질도 이론적으로 고안되고 있으나[96] 임상적으로 확립된 촉진제 후보는 없다. 한편 마취제 및 진정제는 특이한 경우인데 케타민/데스메데토미딘 등의 약물이 유도하는 얕은 마취는 수면과 유사하게 느린파 뇌 활동을 증가시켜 글림파틱 유동을 높인다. 이러한 원리를 이용하여 집중 치료가 필요한 환자들에게 밤 시간에 덱스메데토미딘 등의 진정제를 투여하여 의도적으로 CSF 흐름을 증진시키는 임상 연구도 있다(NCT07151716) [97-99]. 다만 진정의 장기 효과와 안전성 문제로 실제 임상 적용은 신중해야 한다.
이외에도 약물 전달 측면에서 글림파틱 경로를 이용하려는 시도가 있다. 글림파틱 계통이 뇌 곳곳으로 물질을 퍼뜨릴 수 있다는 점에 착안하여 약물을 CSF에 주입한 후 자세 변화나 일주기 리듬을 활용하여 뇌 내 확산을 극대화하는 전략이다. 예를 들어 반감기가 짧은 치료용 단백질이나 안티센스올리고뉴클레오타이드(antisense oligonucleotide) 등을 경막 외 또는 뇌실 내 투여한 뒤 환자를 머리를 낮춘 자세(트렌델렌부르크 자세[Trendelenburg position])로 눕히거나 수면 시간을 이용하여 약물이 뇌조직 깊숙이 전달되게 하는 방식이 연구되고 있다[100,101]. 또한 약물 투여를 야간에 시행하면 주간보다 글림파틱 분포가 더 넓고 뇌 침투가 증가한다는 보고도 있어[44,102] 향후 뇌질환 약물의 투약 시간 조절(time-of-day administration)이 고려될 수 있다.
뇌 글림파틱 계통의 발견과 수막 림프계의 재발견은 CSF 순환에 대한 패러다임 전환을 가져왔다. 이제 우리는 뇌에서 대사 노폐물이 제거되는 경로가 단순 확산이나 정맥 흡수만이 아니라 별아교세포와 PVS를 통한 능동적 유체 교환과 림프관 네트워크에 의해 이루어짐을 인식하게 되었다. 이러한 지식은 알츠하이머병, 파킨슨병 등과 같은 신경퇴행질환부터 뇌졸중, TBI, 소혈관질환 등에 이르기까지 다양한 신경질환에서 수면, 노폐물 축적, 신경 염증 사이의 연결고리를 설명해 준다. 최신 영상을 통해 글림파틱 기능을 측정하고 이를 개선하는 치료법을 탐색하는 연구가 속속 등장하는 만큼 머지않아 임상 신경과 현장에서 글림파틱 건강도를 평가하고 표적화하는 시대가 올 것으로 전망된다. 궁극적으로 수면 위생 개선, 생활 습관 교정, 교감신경 조절 및 림프계 표적 치료 등 다각적 접근을 통해 뇌의 하수도인 글림파틱 계통을 활성화함으로써 신경퇴행질환이나 기타 뇌질환의 예방과 치료에 새로운 장을 열 수 있을 것이다.
Notes
ACKNOWLEDGEMENTS
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. RS-2023-00209580).
REFERENCES
1. Sakka L, Coll G, Chazal J. Anatomy and physiology of cerebrospinal fluid. Eur Ann Otorhinolaryngol Head Neck Dis 2011;128:309-316.


2. Pollay M, Curl F. Secretion of cerebrospinal fluid by the ventricular ependyma of the rabbit. Am J Physiol 1967;213:1031-1038.


3. Oshio K, Watanabe H, Song Y, Verkman AS, Manley GT. Reduced cerebrospinal fluid production and intracranial pressure in mice lacking choroid plexus water channel Aquaporin-1. Faseb J 2005;19:76-78.


4. Bajda J, Pitla N, Gorantla VR. Bulat-Klarica-Oreskovic hypothesis: a comprehensive review. Cureus 2023;15:e45821.



5. Proulx ST. Cerebrospinal fluid outflow: a review of the historical and contemporary evidence for arachnoid villi, perineural routes, and dural lymphatics. Cell Mol Life Sci 2021;78:2429-2457.



6. Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci Transl Med 2012;4:147ra111.



7. Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, et al. Structural and functional features of central nervous system lymphatic vessels. Nature 2015;523:337-341.



8. Absinta M, Ha SK, Nair G, Sati P, Luciano NJ, Palisoc M, et al. Human and nonhuman primate meninges harbor lymphatic vessels that can be visualized noninvasively by MRI. eLife 2017;6:e29738.



9. Albayram MS, Smith G, Tufan F, Tuna IS, Bostancıklıoğlu M, Zile M, et al. Non-invasive MR imaging of human brain lymphatic networks with connections to cervical lymph nodes. Nat Commun 2022;13:203.



10. Ringstad G, Eide PK. Cerebrospinal fluid tracer efflux to parasagittal dura in humans. Nat Commun 2020;11:354.



11. Ahn JH, Cho H, Kim JH, Kim SH, Ham JS, Park I, et al. Meningeal lymphatic vessels at the skull base drain cerebrospinal fluid. Nature 2019;572:62-66.


12. Yoon JH, Jin H, Kim HJ, Hong SP, Yang MJ, Ahn JH, et al. Nasopharyngeal lymphatic plexus is a hub for cerebrospinal fluid drainage. Nature 2024;625:768-777.



13. Redzic ZB, Segal MB. The structure of the choroid plexus and the physiology of the choroid plexus epithelium. Adv Drug Deliv Rev 2004;56:1695-1716.


14. Mollanji R, Bozanovic-Sosic R, Silver I, Li B, Kim C, Midha R, et al. Intracranial pressure accommodation is impaired by blocking pathways leading to extracranial lymphatics. Am J Physiol Regul Integr Comp Physiol 2001;280:R1573-R1581.


15. Albargothy NJ, Johnston DA, MacGregor-Sharp M, Weller RO, Verma A, Hawkes CA, et al. Convective influx/glymphatic system: tracers injected into the CSF enter and leave the brain along separate periarterial basement membrane pathways. Acta Neuropathol 2018;136:139-152.



16. Klarica M, Oresković D, Bozić B, Vukić M, Butković V, Bulat M. New experimental model of acute aqueductal blockage in cats: effects on cerebrospinal fluid pressure and the size of brain ventricles. Neuroscience 2009;158:1397-1405.


17. Bering EA Jr. Choroid plexus and arterial pulsation of cerebrospinal fluid: demonstration of the choroid plexuses as a cerebrospinal fluid pump. AMA Arch Neurol Psychiatry 1955;73:165-172.

19. Dreha-Kulaczewski S, Joseph AA, Merboldt KD, Ludwig HC, Gärtner J, Frahm J. Inspiration is the major regulator of human CSF flow. J Neurosci 2015;35:2485-2491.



20. Yamada S, Miyazaki M, Yamashita Y, Ouyang C, Yui M, Nakahashi M, et al. Influence of respiration on cerebrospinal fluid movement using magnetic resonance spin labeling. Fluids Barriers CNS 2013;10:36.



21. Enzmann DR, Pelc NJ. Normal flow patterns of intracranial and spinal cerebrospinal fluid defined with phase-contrast cine MR imaging. Radiology 1991;178:467-474.

22. van Veluw SJ, Hou SS, Calvo-Rodriguez M, Arbel-Ornath M, Snyder AC, Frosch MP, et al. Vasomotion as a driving force for paravascular clearance in the awake mouse brain. Neuron 2020;105:549-561.e5.


23. Mestre H, Tithof J, Du T, Song W, Peng W, Sweeney AM, et al. Flow of cerebrospinal fluid is driven by arterial pulsations and is reduced in hypertension. Nat Commun 2018;9:4878.


24. Fultz NE, Bonmassar G, Setsompop K, Stickgold RA, Rosen BR, Polimeni JR, et al. Coupled electrophysiological, hemodynamic, and cerebrospinal fluid oscillations in human sleep. Science 2019;366:628-631.


25. Johnston M, Zakharov A, Koh L, Armstrong D. Subarachnoid injection of microfil reveals connections between cerebrospinal fluid and nasal lymphatics in the non-human primate. Neuropathol Appl Neurobiol 2005;31:632-640.


26. Zhang Y, Zhang C, He XZ, Li ZH, Meng JC, Mao RT, et al. Interaction between the glymphatic system and α-synuclein in Parkinson's disease. Mol Neurobiol 2023;60:2209-2222.


27. Nielsen S, Nagelhus EA, Amiry-Moghaddam M, Bourque C, Agre P, Ottersen OP. Specialized membrane domains for water transport in glial cells: high-resolution immunogold cytochemistry of aquaporin-4 in rat brain. J Neurosci 1997;17:171-180.



28. Kress BT, Iliff JJ, Xia M, Wang M, Wei HS, Zeppenfeld D, et al. Impairment of paravascular clearance pathways in the aging brain. Ann Neurol 2014;76:845-861.



29. Zeppenfeld DM, Simon M, Haswell JD, D'Abreo D, Murchison C, Quinn JF, et al. Association of perivascular localization of aquaporin-4 with cognition and Alzheimer disease in aging brains. JAMA Neurol 2017;74:91-99.


30. Zhu YC, Tzourio C, Soumaré A, Mazoyer B, Dufouil C, Chabriat H. Severity of dilated Virchow-Robin spaces is associated with age, blood pressure, and MRI markers of small vessel disease: a population-based study. Stroke 2010;41:2483-2490.


31. Doubal FN, MacLullich AM, Ferguson KJ, Dennis MS, Wardlaw JM. Enlarged perivascular spaces on MRI are a feature of cerebral small vessel disease. Stroke 2010;41:450-454.


33. Banerjee G, Kim HJ, Fox Z, Jäger HR, Wilson D, Charidimou A, et al. MRI-visible perivascular space location is associated with Alzheimer's disease independently of amyloid burden. Brain 2017;140:1107-1116.


34. Martinez-Ramirez S, van Rooden S, Charidimou A, van Opstal AM, Wermer M, Gurol ME, et al. Perivascular spaces volume in sporadic and hereditary (Dutch-type) cerebral amyloid angiopathy. Stroke 2018;49:1913-1919.



35. Charidimou A, Meegahage R, Fox Z, Peeters A, Vandermeeren Y, Laloux P, et al. Enlarged perivascular spaces as a marker of underlying arteriopathy in intracerebral haemorrhage: a multicentre MRI cohort study. J Neurol Neurosurg Psychiatry 2013;84:624-629.



36. Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M, et al. Sleep drives metabolite clearance from the adult brain. Science 2013;342:373-377.



37. Iliff JJ, Lee H, Yu M, Feng T, Logan J, Nedergaard M, et al. Brainwide pathway for waste clearance captured by contrast-enhanced MRI. J Clin Invest 2013;123:1299-1309.



38. Smith AJ, Yao X, Dix JA, Jin BJ, Verkman AS. Test of the 'glymphatic' hypothesis demonstrates diffusive and aquaporin-4-independent solute transport in rodent brain parenchyma. Elife 2017;6:e27679.



39. Ray L, Iliff JJ, Heys JJ. Analysis of convective and diffusive transport in the brain interstitium. Fluids Barriers CNS 2019;16:6.



40. Mestre H, Mori Y, Nedergaard M. The brain’s glymphatic system: current controversies. Trends Neurosci 2020;43:458-466.



41. Ma J, Chen M, Liu GH, Gao M, Chen NH, Toh CH, et al. Effects of sleep on the glymphatic functioning and multimodal human brain network affecting memory in older adults. Mol Psychiatry 2025;30:1717-1729.


42. Ghaderi S, Mohammadi S, Fatehi F. Glymphatic pathway dysfunction in severe obstructive sleep apnea: a meta-analysis. Sleep Med 2025;131:106528.


43. Ozturk B, Koundal S, Al Bizri E, Chen X, Gursky Z, Dai F, et al. Continuous positive airway pressure increases CSF flow and glymphatic transport. JCI Insight 2023;8:e170270.



44. Hablitz LM, Plá V, Giannetto M, Vinitsky HS, Stæger FF, Metcalfe T, et al. Circadian control of brain glymphatic and lymphatic fluid flow. Nat Commun 2020;11:4411.



46. Hussain R, Tithof J, Wang W, Cheetham-West A, Song W, Peng W, et al. Potentiating glymphatic drainage minimizes post-traumatic cerebral oedema. Nature 2023;623:992-1000.


47. Cheng KP, Brodnick SK, Blanz SL, Zeng W, Kegel J, Pisaniello JA, et al. Clinically-derived vagus nerve stimulation enhances cerebrospinal fluid penetrance. Brain Stimul 2020;13:1024-1030.


48. Kiviniemi V, Wang X, Korhonen V, Keinänen T, Tuovinen T, Autio J, et al. Ultra-fast magnetic resonance encephalography of physiological brain activity - Glymphatic pulsation mechanisms? J Cereb Blood Flow Metab 2016;36:1033-1045.


49. Yildiz S, Grinstead J, Hildebrand A, Oshinski J, Rooney WD, Lim MM, et al. Immediate impact of yogic breathing on pulsatile cerebrospinal fluid dynamics. Sci Rep 2022;12:10894.



50. Iliff JJ, Wang M, Zeppenfeld DM, Venkataraman A, Plog BA, Liao Y, et al. cerebral arterial pulsation drives paravascular CSF-interstitial fluid exchange in the murine brain. J Neurosci 2013;33:18190-18199.



51. Li M, Kitamura A, Beverley J, Koudelka J, Duncombe J, Lennen R, et al. Impaired glymphatic function and pulsation alterations in a mouse model of vascular cognitive impairment. Front Aging Neurosci 2022;13:788519.



52. Jukkola J, Kaakinen M, Singh A, Moradi S, Ferdinando H, Myllylä T, et al. Blood pressure lowering enhances cerebrospinal fluid efflux to the systemic circulation primarily via the lymphatic vasculature. Fluids Barriers CNS 2024;21:12.



53. Xie L, Zhang Y, Hong H, Xu S, Cui L, Wang S, et al. Higher intracranial arterial pulsatility is associated with presumed imaging markers of the glymphatic system: an explorative study. Neuroimage 2024;288:120524.


54. Ringstad G, Vatnehol SAS, Eide PK. Glymphatic MRI in idiopathic normal pressure hydrocephalus. Brain 2017;140:2691-2705.



55. Taoka T, Masutani Y, Kawai H, Nakane T, Matsuoka K, Yasuno F, et al. Evaluation of glymphatic system activity with the diffusion MR technique: diffusion tensor image analysis along the perivascular space (DTI-ALPS) in Alzheimer's disease cases. Jpn J Radiol 2017;35:172-178.


56. Zhang X, Wang Y, Jiao B, Wang Z, Shi J, Zhang Y, et al. Glymphatic system impairment in Alzheimer's disease: associations with perivascular space volume and cognitive function. Eur Radiol 2024;34:1314-1323.


57. Liang T, Chang F, Huang Z, Peng D, Zhou X, Liu W. Evaluation of glymphatic system activity by diffusion tensor image analysis along the perivascular space (DTI-ALPS) in dementia patients. Br J Radiol 2023;96:20220315.



58. Bae YJ, Kim JM, Choi BS, Choi JH, Ryoo N, Song YS, et al. Glymphatic function assessment in Parkinson's disease using diffusion tensor image analysis along the perivascular space. Parkinsonism Relat Disord 2023;114:105767.


59. Wood KH, Nenert R, Miften AM, Kent GW, Sleyster M, Memon RA, et al. Diffusion tensor imaging-along the perivascular-space index is associated with disease progression in Parkinson's disease. Mov Disord 2024;39:1504-1513.


60. Yu S, Jiang H, Yu L, Liu T, Yang C, Cao J, et al. DTI-ALPS index decreased in patients with type 2 diabetes mellitus. Front Neurosci 2024;18:1383780.



61. Tian Y, Cai X, Zhou Y, Jin A, Wang S, Yang Y, et al. Impaired glymphatic system as evidenced by low diffusivity along perivascular spaces is associated with cerebral small vessel disease: a population-based study. Stroke Vasc Neurol 2023;8:413-423.


62. Ringstad G. Glymphatic imaging: a critical look at the DTI-ALPS index. Neuroradiology 2024;66:157-160.


63. Potter GM, Chappell FM, Morris Z, Wardlaw JM. Cerebral perivascular spaces visible on magnetic resonance imaging: development of a qualitative rating scale and its observer reliability. Cerebrovasc Dis 2015;39:224-231.



64. Dubost F, Yilmaz P, Adams H, Bortsova G, Ikram MA, Niessen W, et al. Enlarged perivascular spaces in brain MRI: automated quantification in four regions. Neuroimage 2019;185:534-544.

65. Kapoor A, Gaubert A, Yew B, Jang JY, Dutt S, Li Y, et al. Enlarged perivascular spaces and plasma Aβ42/Aβ40 ratio in older adults without dementia. Neurobiol Aging 2023;128:43-48.



66. Jeong SH, Cha J, Park M, Jung JH, Ye BS, Sohn YH, et al. Association of enlarged perivascular spaces with amyloid burden and cognitive decline in Alzheimer disease continuum. Neurology 2022;99:e1791-e1802.


67. Kim S, Na HK, Sun Y, Yoon YJ, Chung SJ, Sohn YH, et al. Regional burden of enlarged perivascular spaces and cognition and neuropsychiatric symptoms in drug-naive patients with Parkinson disease. Neurology 2024;102:e209483.


68. Park YW, Shin NY, Chung SJ, Kim J, Lim SM, Lee PH, et al. Magnetic resonance imaging-visible perivascular spaces in basal ganglia predict cognitive decline in Parkinson's disease. Mov Disord 2019;34:1672-1679.


69. Chung SJ, Yoo HS, Shin NY, Park YW, Lee HS, Hong JM, et al. Perivascular spaces in the basal ganglia and long-term motor prognosis in newly diagnosed Parkinson disease. Neurology 2021;96:e2121-e2131.


70. Yamada S, Hiratsuka S, Otani T, Ii S, Wada S, Oshima M, et al. Usefulness of intravoxel incoherent motion MRI for visualizing slow cerebrospinal fluid motion. Fluids Barriers CNS 2023;20:16.



71. Chen Y, Dai Z, Fan R, Mikulis DJ, Qiu J, Shen Z, et al. Glymphatic system visualized by chemical-exchange-saturation-transfer magnetic resonance imaging. ACS Chem Neurosci 2020;11:1978-1984.


72. Boyd ED, Kaur J, Ding G, Chopp M, Jiang Q. Clinical magnetic resonance imaging evaluation of glymphatic function. NMR Biomed 2024;37:e5132.


73. Zhou Y, Cai J, Zhang W, Gong X, Yan S, Zhang K, et al. Impairment of the glymphatic pathway and putative meningeal lymphatic vessels in the aging human. Ann Neurol 2020;87:357-369.


74. Nedergaard M, Goldman SA. Glymphatic failure as a final common pathway to dementia. Science 2020;370:50-56.



75. Peng W, Achariyar TM, Li B, Liao Y, Mestre H, Hitomi E, et al. Suppression of glymphatic fluid transport in a mouse model of Alzheimer's disease. Neurobiol Dis 2016;93:215-225.



76. Harrison IF, Ismail O, Machhada A, Colgan N, Ohene Y, Nahavandi P, et al. Impaired glymphatic function and clearance of tau in an Alzheimer’s disease model. Brain 2020;143:2576-2593.



77. Kamagata K, Andica C, Takabayashi K, Saito Y, Taoka T, Nozaki H, et al. Association of MRI indices of glymphatic system with amyloid deposition and cognition in mild cognitive impairment and Alzheimer DIsease. Neurology 2022;99:e2648-e2660.



78. Ang PS, Zhang DM, Azizi SA, de Matos SAN, Brorson JR. The glymphatic system and cerebral small vessel disease. J Stroke Cerebrovasc Dis 2024;33:107557.



79. Ding J, Sigurðsson S, Jónsson PV, Eiriksdottir G, Charidimou A, Lopez OL, et al. Large perivascular spaces visible on magnetic resonance imaging, cerebral small vessel disease progression, and risk of dementia: the age, gene/environment susceptibility - Reykjavik study. JAMA Neurol 2017;74:1105-1112.


80. Zhang S, Yu W, Zhang X, Cheng H, Zhang Z, Gao S, et al. Glymphatic dysfunction as a biomarker for post-stroke cognitive impairment. Sci Rep 2025;15:19382.



81. Dumas AA, Gasull AD, Prinz M. Beyond borders: the choroid plexus-immune communication during neuroinflammation. Signal Transduct Target Ther 2024;9:278.



82. Serot JM, Béné MC, Foliguet B, Faure GC. Morphological alterations of the choroid plexus in late-onset Alzheimer's disease. Acta Neuropathol 2000;99:105-108.


83. Li Y, Zhou Y, Zhong W, Zhu X, Chen Y, Zhang K, et al. Choroid plexus enlargement exacerbates white matter hyperintensity growth through glymphatic impairment. Ann Neurol 2023;94:182-195.

84. Xu X, Yang X, Zhang J, Wang Y, Selim M, Zheng Y, et al. Choroid plexus free-water correlates with glymphatic function in Alzheimer's disease. Alzheimers Dement 2025;21:e70239.



85. Liu L, Weng Q, Cai Q, Yu X, Huang W, Xie S, et al. Choroid plexus enlargement contributes to motor severity via regional glymphatic dysfunction in Parkinson’s disease. NPJ Parkinsons Dis 2025;11:134.



86. Jeong SH, Park CJ, Cha J, Kim SY, Lee SK, Kim YJ, et al. Choroid Plexus volume, amyloid burden, and cognition in the Alzheimer's disease continuum. Aging Dis 2025;16:552-564.
87. Jeong SH, Park CJ, Jeong HJ, Sunwoo MK, Ahn SS, Lee SK, et al. Association of choroid plexus volume with motor symptoms and dopaminergic degeneration in Parkinson's disease. J Neurol Neurosurg Psychiatry 2023;94:1047-1055.


88. Lolansen SD, Révész EO, Andreassen SN, Trapp M, Salio C, Sassoé-Pognetto M, et al. Choroid plexus-mediated CSF secretion remains stable in aging rats via high and age-resistant metabolic activity. Nat Commun 2025;16:6778.



89. Nishioka K, Kawamura M, Iima M, Ueda D, Ito R, Saida T, et al. The glymphatic system in oncology: from the perspective of a radiation oncologist. J Radiat Res 2025;66:343-353.



90. Lee H, Xie L, Yu M, Kang H, Feng T, Deane R, et al. The effect of body posture on brain glymphatic transport. J Neurosci 2015;35:11034-11044.



91. Yoo RE, Kim JH, Moon HY, Park JY, Cheon S, Shin HS, et al. Long-term physical exercise facilitates putative glymphatic and meningeal lymphatic vessel flow in humans. Nat Commun 2025;16:3360.



92. Lundgaard I, Wang W, Eberhardt A, Vinitsky HS, Reeves BC, Peng S, et al. Beneficial effects of low alcohol exposure, but adverse effects of high alcohol intake on glymphatic function. Sci Rep 2018;8:2246.



93. Ren H, Luo C, Feng Y, Yao X, Shi Z, Liang F, et al. Omega-3 polyunsaturated fatty acids promote amyloid-β clearance from the brain through mediating the function of the glymphatic system. Faseb J 2017;31:282-293.


94. Zhang Y, Zhang R, Ye Y, Wang S, Jiaerken Y, Hong H, et al. The influence of demographics and vascular risk factors on glymphatic function measured by diffusion along perivascular space. Front Aging Neurosci 2021;13:693787.



95. Orešković D, Maraković J, Varda R, Radoš M, Jurjević I, Klarica M. New insight into the mechanism of mannitol effects on cerebrospinal fluid pressure decrease and craniospinal fluid redistribution. Neuroscience 2018;392:164-171.


96. Mestre H, Hablitz LM, Xavier ALR, Feng W, Zou W, Pu T, et al. Aquaporin-4-dependent glymphatic solute transport in the rodent brain. eLife 2018;7:e40070.


97. Lilius TO, Blomqvist K, Hauglund NL, Liu G, Stæger FF, Bærentzen S, et al. Dexmedetomidine enhances glymphatic brain delivery of intrathecally administered drugs. J Control Release 2019;304:29-38.


98. Dong R, Liu W, Han Y, Wang Z, Jiang L, Wang L, et al. Influencing factors of glymphatic system during perioperative period. Front Neurosci 2024;18:1428085.



99. Hablitz LM, Vinitsky HS, Sun Q, Stæger FF, Sigurdsson B, Mortensen KN, et al. Increased glymphatic influx is correlated with high EEG delta power and low heart rate in mice under anesthesia. Sci Adv 2019;5:eaav5447.



100. Hinderer C, Katz N, Dyer C, Goode T, Johansson J, Bell P, et al. Translational feasibility of lumbar puncture for intrathecal AAV administration. Mol Ther Methods Clin Dev 2020;17:969-974.



Figure 1.
Schematic overview of the glymphatic system. (A) Cerebrospinal fluid (CSF) enters the brain along the periarterial spaces. (B) CSF flow is driven by arterial pulsatility and related forces, and CSF is transported into the brain parenchyma through aquaporin-4 water channels that are highly enriched in astrocytic endfeet. (C) Within the brain tissue, CSF and interstitial fluid mix via diffusion and convection, facilitating the movement and clearance of metabolic waste products. (D) Drainage occurs predominantly along white matter axonal tracts and perivenous spaces, allowing waste to be conveyed toward the parasagittal region and venous sinuses into the subarachnoid space. (E) Subsequently, waste products exit the cranial cavity via meningeal lymphatic vessels or arachnoid granulations, or (F) along perineural pathways associated with cranial nerves.
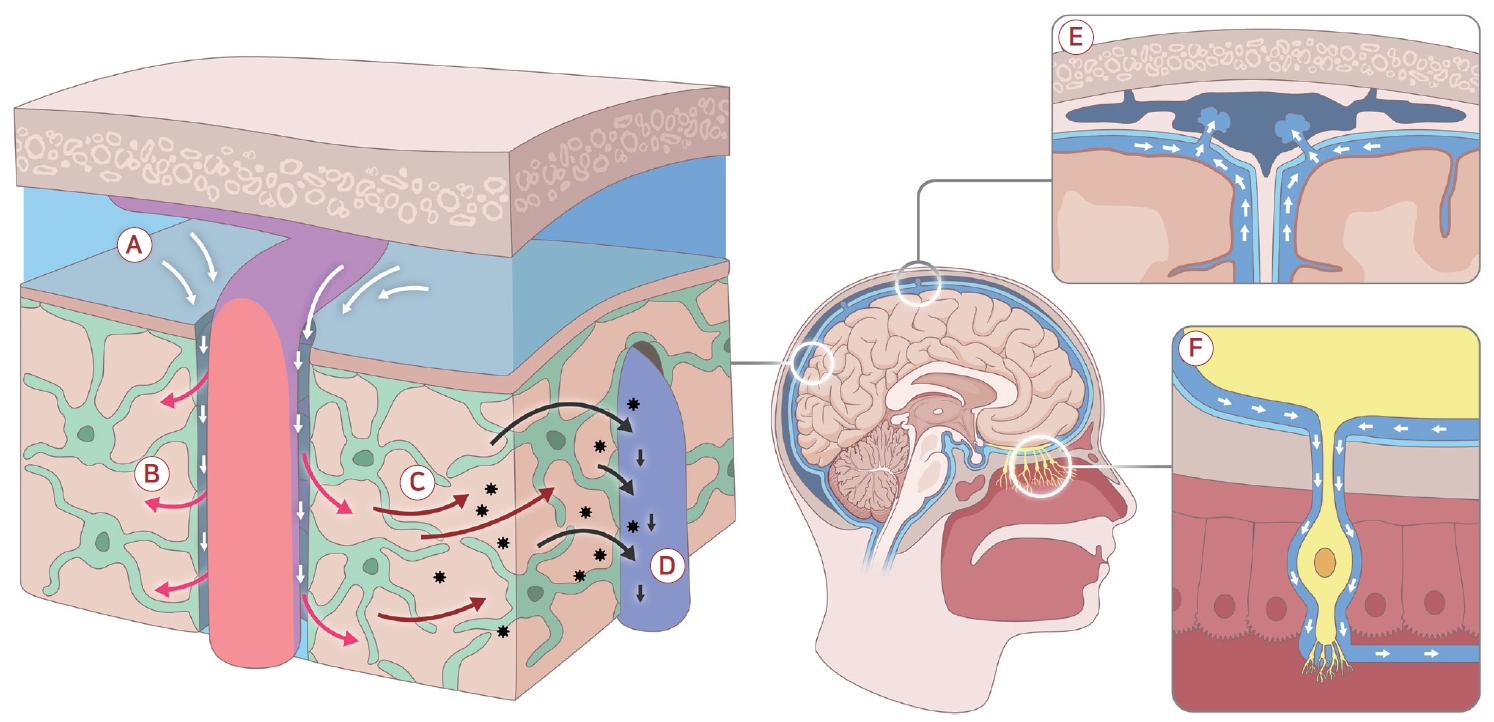
Table 1.
Comparison of diffusion- and convection-based mechanisms in the glymphatic pathway [40]
- TOOLS



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print